Maximization of entropy sets fundamental limits on the amount of useful work that a process can perform. When is entropy production maximized in the active biological matter, and what does it say about its mechanics?
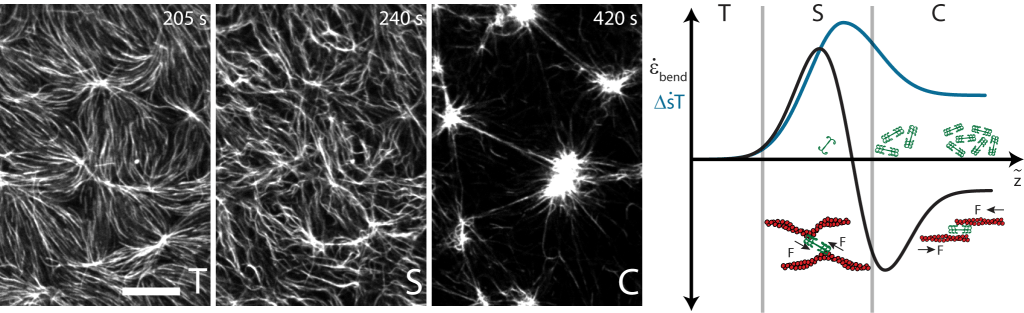
In work published in the journal Nature Communications, We estimate the mechanical dissipation in contractile actomyosin by enumerating an actin network’s configurational and binding entropy. We show that the balance of work and entropy does not increase monotonically, and the entropy production rate is maximum in the non-contractile, stable state of actomyosin. Our study provides evidence that the origins of entropy production and activity-dependent dissipation relate to disorder in the molecular interactions between actin and myosin.
Unlike nearly all engineered materials that contain bonds that weaken under load, cytoskeletal networks contain “catch” bonds that are reinforced under load. Consequently, materials such as the actomyosin networks can adapt their mechanical properties in response to their state of internal, non-equilibrium (active) stress. In work published in the journal Advanced Functional Materials, through coarse-grained molecular dynamics simulations, we explore the effect of the local breaking of detailed balance, as a measure of distance from equilibrium, on the accumulation and dissipation of energy within a model of the actomyosin cytoskeleton. We find that the extent to which detailed balance is broken uniquely determines a large-scale fluid-solid transition with characteristic time-reversal symmetries.
Representative publications:
“Entropy production rate is maximized in non-contractile actomyosin,” D. S. Seara, V. Yadav, I. Linsmeier, A. P. Tabatabai, P. W. Oakes, SM Ali Tabei, S. Banerjee, M. P. Murrell, Nature Communications (2018).
“Detailed balance broken by catch bond kinetics enables mechanical‐adaptation in active materials,” A. P. Tabatabai, D. S. Seara, J. Tibbs, V. Yadav, I. Linsmeier, M. P. Murrell, Advanced Functional Materials (2020).
You must be logged in to post a comment.