There are fundamental laws that govern the behavior of physical systems. For example, Newton’s laws define the motion of a classical particle, whereas the Navier-Stokes equation defines the motion of a fluid. Are there any fundamental principles in biology that dictate the growth and motion of a tumor or tissue?
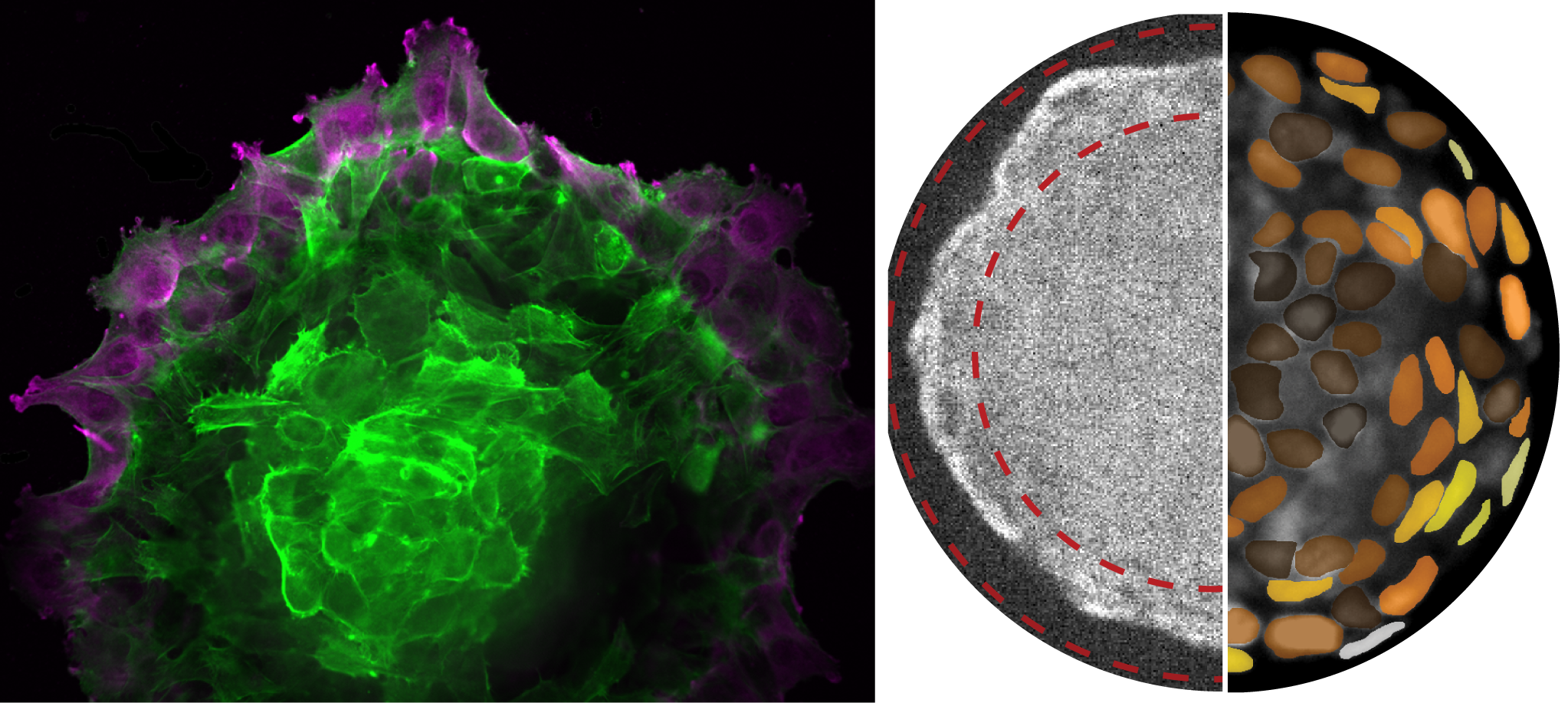
There are no known equations for the growth of a tumor as methods and models developed for equilibrium systems do not apply to a tumor that is inherently out of equilibrium. In our research, we use cellular aggregates as simplified models of tissues and tumors. Using an extensive series of experimental measurements, agent-based simulations, and a non-equilibrium thermodynamic framework, we have proposed a set of equations quantifying a tumor’s growth and spreading rate. We find that cellular motion inside a tumor generates active stresses, which lead to surface tension and cellular density of the tumor becoming size-dependent. These quantities are well-defined constants for usual materials but vary with size in a tumor. However, a carefully chosen combination of surface tension and density uniquely identifies the state of the tissue. Upon wetting of tumors on compliant substrates, this size-dependent surface tension competes with the stiffness of the substrate and determines the spreading rate of the tumor. As surface tension also dictates the tumor’s internal pressure, small tumors on soft substrates spread exponentially fast using a pressure-driven mechanism, whereas tumors on stiff substrates spread only linearly fast using a traction-driven mechanism. Surprisingly, the length scale over which the spreading behavior of the tumor changes from a pressure-driven to a traction-driven mechanism is defined by its elastocapillary length.
Representative publications:
“Cell-Matrix elastocapillary interactions drive pressure-based wetting of cell aggregates,” M.S. Yousafzai*, V. Yadav*, S. Amiri, M.F. Staddon, Y. Errami, G. Jaspard, S. Banerjee, and M.P. Murrell, Physical Review X (2022).
“Gradients in solid surface tension drive Marangoni-like motions in cell aggregates,” V. Yadav*, M.S. Yousafzai*, S. Amiri, Y. Errami, S. Amiri, R.W. Style, E.R. Dufresne, and M.P. Murrell, Physical Review Fluids, (2022).“Active Regulation of Pressure and Volume Defines an Energetic Constraint on the Size of Cell Aggregates,” M.S. Yousafzai*, V. Yadav*, S. Amiri, Y. Errami, S. Amiri, and M.P. Murrell, Physical Review Letters, (2022).
“Tissue pressure and cell traction compensate to drive robust aggregate spreading,” Md S. Yousafzai*, V. Yadav*, S. Amiri, M. Staddon, A. Pasha Tabatabai, Y. Errami, G. Jaspard, S. Amiri, S. Banerjee, and M. P. Murrell, BioRxiv, (2020)
You must be logged in to post a comment.